
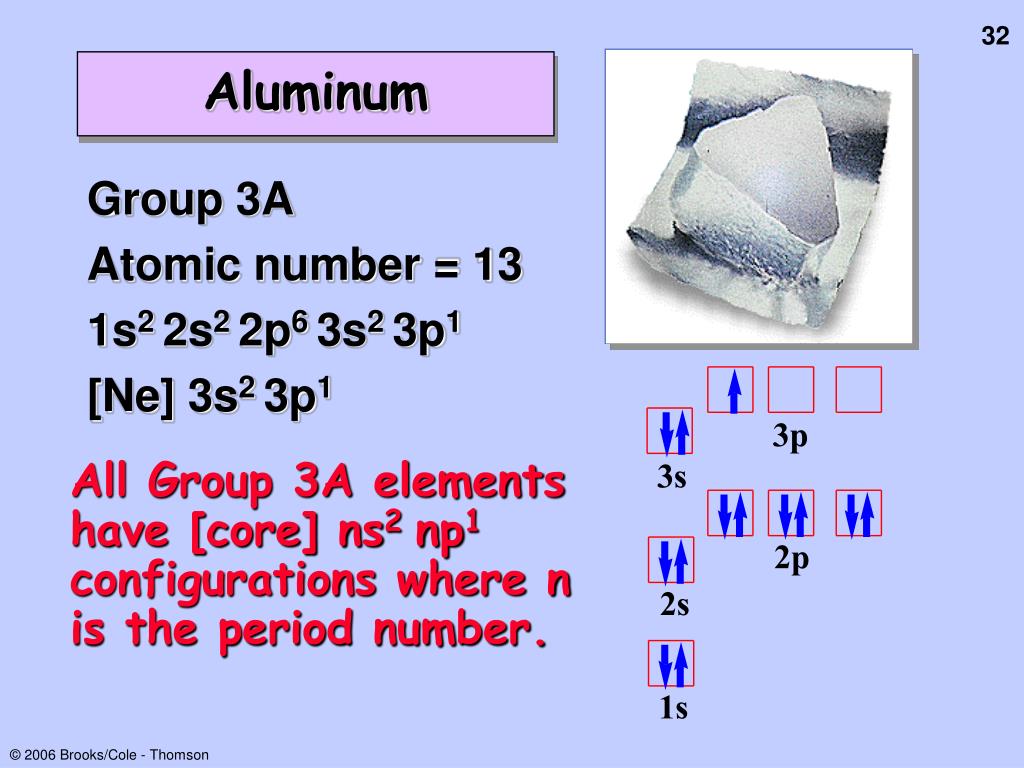
of s 1 electrons 1s label of l value of n for H, atomic number = 1 Orbital Box notation spdf notation 1 no. Electron configuration can be given with the orbital box diagram, or with the spdf notation.In general, electrons are assigned to orbitals in order of increasing _.The arrangements of _ in the elements in the ground state.2s electron experiences a _ positive charge than expected.The 2s electron PENETRATES the region occupied by the 1s electron.– s electrons always have a lower energy than p electrons in the same quantum shell. Z* is _ for s electrons than for p electrons.C has 6 p (+) and 6 e (-) 2 e- in 1s 2 e- in 2s 2 e- in 2p e- in 2s should “see” +3, but see 3.22 e- in 2p should “see” a +2 charge, but see 3.14.Li has 3 p (+) and 3 e (-) 2 e in 1 s orbital 1 e in 2 s orbital e- in 2s should “see” a +1 charge, but it sees 1.28.Z* - the nuclear charge experienced by a particular electron in a multielectron atom, as modified by the presence of the other electrons.1 e- atomĮlectron cloud for 1s electrons Effective Nuclear Charge, Z* b) for subshells of same _, subshell with lower n is lower in energy. In many-electron atom: a) subshells increase in energy as value of _increases.For many-electron atoms, energy depends on both _ and _.Electrons generally assigned to orbitals of successively higher energy.3Ē +1 +1/2 Students should be familiar with the values and meaning of quantum numbers. Ī 2p electron can be designated by which set of quantum numbers? n l ml ms a. this shell has a single orbital (1s) to which 2e- can be assigned.Therefore, Each orbital can be assigned no more than _ electrons! The Pauli Exclusion Principle No two electrons in an atom can have the same set of four quantum numbers.

ms, _ quantum number, = +1/2 and -1/2.Understand the fundamental physical properties of the elements and their periodic trends. Write the electron configuration for elements and monoatomic ions.

Understand effective nuclear charge and its role in determining atomic properties. Atomic Electron Configurations and Chemical Periodicity Goals: Understand the role magnetism plays in determining and revealing atomic structure.

